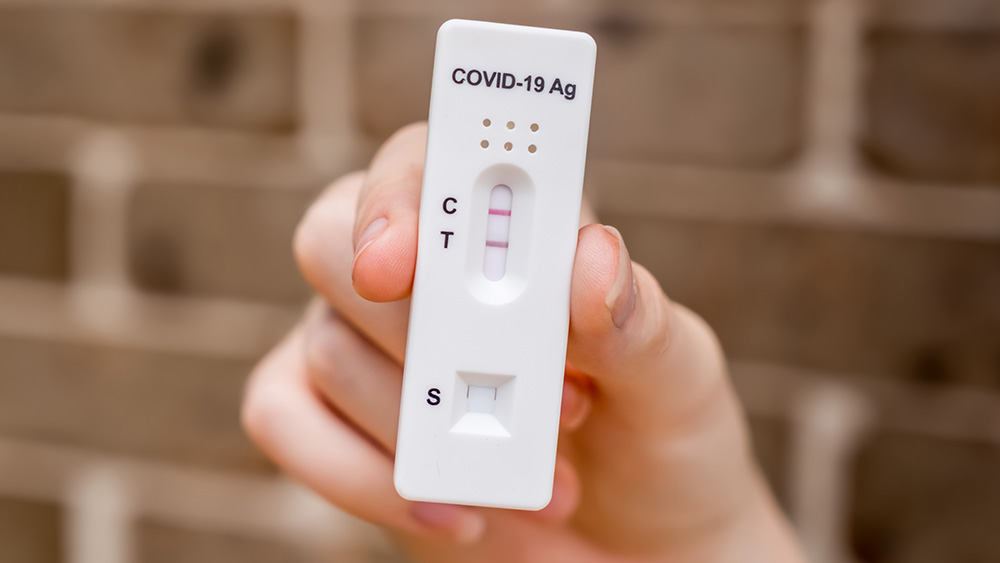
MakeMyTestCount.org
At MakeMyTestCount, you can report the results of your at-home COVID-19 test safely and anonymously. Public health officials can use this information to keep our communities safe.

COVID-19 Testing Basics
How do I get tested?
Most health care providers offer COVID-19 testing and should be contacted first if you are experiencing symptoms. Your state health department’s website will have information on additional local testing sites.
You can also schedule an appointment for a COVID-19 diagnostic test at many national pharmacy chains. Stores with drive-through lanes will ask you to remain in your car while a pharmacist guides you through the self-testing processes. Results are usually available in a few days.
How can I get an at-home test?
At-home rapid tests can be purchased over the counter at many pharmacies, with results ready in as little as 15 minutes. The CDC has more information about self-testing, including how to swab yourself.
Residential households in the U.S. can order free at-home tests from the U.S. Postal Service. Visit covidtests.gov to place an order.
NIH has created MakeMyTestCount, a site that allows you to report your at-home COVID-19 test results safely and privately. Public health officials can use this anonymous data to track COVID-19 levels in our communities and keep us safe. This project is funded through the NIH RADx® Tech Initiative.
More information on at-home tests and testing:
-
The U.S. Food and Drug Administration (FDA) maintains a list of authorized at-home OTC COVID-19 diagnostic tests. The list also shows updated information about the shelf life and expiration dates of these tests.
-
The FDA has recalled Celltrion DiaTrust COVID-19 Ag Rapid Test (labeled for Research Use Only) over labeling issues. You can avoid getting a false result by not using these tests.
What should I do if I test positive for COVID-19?
Most people with COVID-19 have mild illness and can recover at home. You can help yourself recover and keep the virus from spreading within your family and to others by following the CDC guidelines for at-home COVID-19 care. If you are experiencing moderate to severe COVID-19 symptoms, talk to your doctor.
In March 2022, the White House launched the Test to Treat initiative to give people quick access to life-saving COVID-19 treatments. Hundreds of participating pharmacy-based clinics, community health centers, and long-term care facilities now provide free testing for COVID-19 and allow a person who tests positive at their site to be evaluated by a health care professional. If the treatments are right for them, they can be given antiviral medication right away. Find your closest Test to Treat site here or call 1-800-232-0233.
When should I get tested for COVID-19?
If you have been exposed to COVID-19, you should get tested — even if you have been vaccinated. Get tested for COVID-19:
-
If you develop COVID-19 symptoms
-
At least 5 days after known or suspected exposure to someone with COVID-19
What are the types of COVID-19 tests?
A viral test looks for current infection. There are two types of viral tests:
-
The antigen test (also called the “rapid test”) gives results in as little as 15 minutes. You are more likely to get a false negative result from the antigen test. If you have symptoms but get a negative test result from the antigen test, your doctor may give you a molecular test to confirm the results.
-
The molecular test can take a few days to a week to provide results. The most common test relies on a laboratory method called polymerase chain reaction (PCR) to detect SARS-CoV-2 genetic material.
An antibody test is designed to tell you whether you had an infection in the past.


The State of COVID-19 Testing
Why do we still need testing?
Testing is critical to controlling the spread of SARS-CoV-2, the virus that causes COVID-19. Testing is the only way to be sure you are not passing the virus on to others — even people who are vaccinated can carry the virus while showing few to no symptoms.
Testing also provides important information about the virus’s movement within and between communities. Public health officials need to know how many people are infected and where they live so that the officials can track outbreaks and react accordingly.
How is NIH continuing to support and improve testing?
At the beginning of the pandemic, NIH moved quickly to support the development of fast and accurate testing, awarding nearly half a billion dollars through the RADx® initiative to help rapidly scale up the country’s testing capacity.
Additional grants and research continue to support efforts to create easy-to-use tests, implement innovative ways of getting people tested, and reach underserved communities to ensure that anyone who needs a test can easily get one.
What is the future of testing?
NIH will continue to support research to make testing easier and more efficient. New testing devices and protocols are being developed and studied every day.
COVID-19 Testing Resources
The CDC shares information about testing and a COVID-19 symptoms checker.
The U.S. Food and Drug Administration shares facts about testing regulation and approval.
The U.S. Department of Health and Human Services (HHS) shares information about where and how to get tested.
The RADx-UP initiative provides communications toolkits in English and Spanish, including resources developed for American Indian tribes.
 An official website of the United States government
An official website of the United States government